Electrostatic Repulsion Hydrophilic Interaction Chromatography
Or in short “ERLIC”. Electrostatic Repulsion Hydrophilic Interaction Chromatography is a new mode of chromatography. It permits some separations to be done isocratically that normally would require a gradient. It also permits the selective isolation of phosphopeptides from a tryptic digest.
ERLIC, the column is of the same charge as the sample solutes. The mobile phase contains enough organic solvent so that hydrophilic interaction keeps the solutes on the column despite the electrostatic repulsion. The pH of the mobile phase is selected to insure that the solutes do have the same charge as the column. When the best-retained solutes in a mixture are highly charged, this combination can be used to selectively antagonize their retention.
To separate nucleotides and nucleic acids use cation-exchange columns such as PolySULFOETHYL A or PolyCAT A. The solutes are negatively charged.
Separating for amino acids, peptides and proteins use Anion-exchange columns such as PolyWAX LP
ERLIC vs HILIC with Peptides
The example below shows the elution of a mixture of acidic, basic and neutral peptides
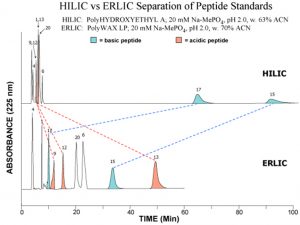
TOP: HILIC mode on a PolyHYDROXYETHYL A column. The basic peptides are much better retained than the neutral or basic peptides, since basic solutes are the most polar of all.
BOTTOM: ERLIC mode on a PolyWAX LP column. Selective repulsion of the basic peptides throws them into the same elution time frame as the other peptides.
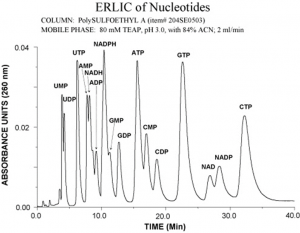
Electrostatic Repulsion Hydrophilic Interaction Chromatography of Nucleotides
The following example shows the separation of nucleotides on PolySULFOETHYL A. Normally the NTP’s elute much later than the NMP’s and NDP’s in either the anion-exchange or HILIC mode. In ERLIC they elute isocratically in the same time frame as the other nucleotides.
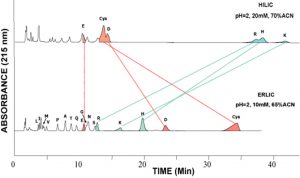
ERLIC vs HILIC of Amino Acids
This example shows the separation of amino acids on a PolyHYDROXYETHYL A column in the HILIC mode [top] and on a PolyWAX LP column in the ERLIC mode [bottom]. Basic amino acids are much better retained than the others in HILIC but not in ERLIC.
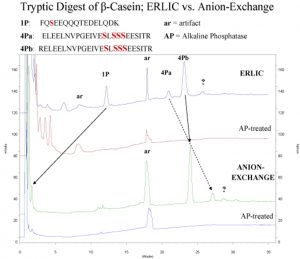
Electrostatic Repulsion Hydrophilic Interaction Chromatography of Phosphopeptides:
ERLIC of peptides is normally performed with a PolyWAX LP column at a pH ~ 2.0, low enough to uncharge carboxyl- groups in peptides. Under these conditions, phosphate groups retain their negative charge and are electrostatically attracted to the column. This attraction is not sufficient to overcome the electrostatic repulsion from the two basic groups in a typical tryptic peptide (the N-terminus and the Lys- or Arg- residue at the C-terminus), which is why anion-exchange has not been used for selective isolation of phosphopeptides from tryptic digests. Phosphate groups are quite hydrophilic, though. In the ERLIC mode the combination of electrostatic attraction and hydrophilic interaction does suffice to pull phosphopeptides away from the nonphosphorylated peptides in a tryptic digest. This is clear from the following example, which contrasts retention of tryptic peptides from beta-casein on the same column in the ERLIC mode and in the anion-exchange mode.
In addition to isolating the phosphopeptides from the nonphosphopeptides in tryptic digests, ERLIC is also a high-resolution method for separating the phosphopeptides from each other. Analysis of a complex digest via ERLIC can easily result in the identification of > 2000 phosphopeptides via bottom-up proteomics methods. Distribution of these phosphopeptides into 20-40 fractions facilitates their identification. Peptides with more than one phosphate group are so strongly retained in ERLIC that a salt gradient is necessary for their elution. If nonvolatile salts are used, then the collected fractions have to be desalted prior to further analysis. The material used for the purpose is important, since many phosphopeptides are too polar to be retained by C-18 material. The following figure compares desalting with HyperCarb vs. desalting with C-18:
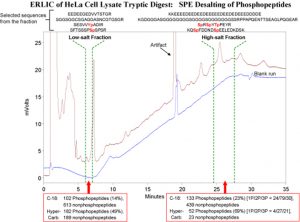
Observations:
1) Low-salt fraction: HyperCarb retains ~ 50% more phosphopeptides than does C-18, and appreciably less nonphosphopeptides.
2) High-salt fraction: C-18 retains more peptides of all kinds, phosphorylated and nonphosphorylated.
3) The peptides in the late-eluting fraction tend to have 2 or 3 phosphate groups. The peptide sequences displayed are reasonably characteristic of the types of peptides observed in the fractions in question.
4) Neither material captured all of the phosphopeptides that were retained by the other material in either case, although there was considerable overlap.
Conclusions:
Use HyperCarb SPE cartridges (e.g., our TopTips) to desalt phosphopeptides in low-salt media (< 60 mM). In high-salt media (> 100 mM), use C-18 SPE cartridges. For maximum identification of phosphopeptides, use both materials in parallel for each sample and then either pool the resulting eluates or else proceed to analyze them in parallel.
Literature
http://www.polylc.com/Downloads/Poster_ASMS_2008_ERLIC_of_phosphopeptides.pdf
http://www.polylc.com/Downloads/HPLC_2008_poster_2412-W_ERLIC_of_phosphopeptides_slides.pdf
http://www.polylc.com/Downloads/ERLIC_of_Phosphopeptides_bulletin.pdf
http://www.polylc.com/Downloads/ERLIC_bulletin_6-2007.pdf
http://www.polylc.com/Downloads/ASMS_2007_Desalting_Phosphopeptides_Alpert_et_al.pdf